
Presenter: Robert Lutskus, Associate Director Commercial Operations at Lonza
Rob was a Quality Control microbiologist for 10 years in pharma before transitioning to the laboratory informatics space. He has run and supported implementations and validations of software at numerous organizations globally and now heads the delivery team for the MODA EM solution.
Overview
Lab instruments and enterprise systems that solve specific needs reside in silos within a manufacturing site.
Connecting these devices and systems provides the ability to capture for better, more efficient decisions, and will error proof the workflow by removing data transcription.
The how, why, when and for whom should organizations connect devices and systems will determine how efficiently and effectively organizations can derive value from the data they generate, and ultimately, error proof workflows and processes.
A thoughtfully designed, connected lab, and connected data add business value by allowing sites to increase capacity.
This session will discuss the value of defining integrations, the technical aspects of having devices and systems exchange data, setting up validation protocols to manage data integrity concerns, and the process improvements that will ultimately be realized.
Why attending
A unique opportunity for all those involved in digitalization journey to learn the value that well-defined integrations can add to your process, and ultimately help release the product faster than ever before.
Who should benefit
This session is appropriate for QC and Manufacturing personnel who generate the data, QA personnel who review and release data, system owners and IT Business partners.
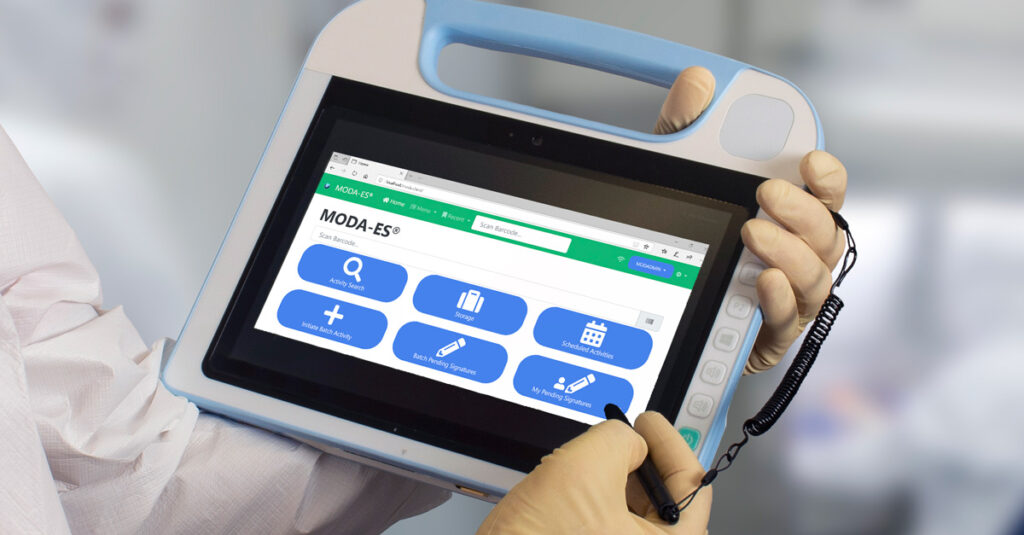

With a deep understanding of the complex challenges faced by the pharmaceutical & biotechnology companies, Lonza Informatics is committed to empowering organizations to accelerate process development, and manufacturing.Our flagship platform, MODA®, is a scalable, platform that centralizes data capture, management & analysis, and compliance in regulated environments.
With the MODA® Platform, small & large scale manufacturing organizations can effectively achieve operational excellence, productivity gains, and ensure regulatory compliance throughout the product life cycle.
Propel your organization towards success, contact Lonza Informatics today to learn more information. www.lonza.com/moda







