Paperless Lab Academy® 2019: Digital Optimization vs Digital Transformation

Paperless Lab Academy®: a point of reference for your laboratory digital transformation projects
The best way to summarize the seventh edition of the Paperless Lab Academy® is the path of the discussions that have evolved from the 20 years history of the laboratory informatics up to the management of our own personal data as citizens and consumers.
The Academy has now become a point of reference for those looking for answers, for those in need for understanding how to implement a Digital Transformation (DT) of their quality processes. The consolidated reputation of the quality of the presentations and the networking opportunities in an environment that has few equals makes the Paperless Lab Academy® one of the best congress in Europe for discussing Lab informatics and scientific data management.
Paperless Lab Academy® 2019: spotlight on …
Starting from Sergio Nasi, Head of IT RDM LIMS, Boehringer Ingelheim, key-note speaker of this edition, the participants ventured into the history of the laboratory informatics. In fact, LIMS has existed since 1982, well before other systems much more known as ERPs. This means that the needs to digitalize the laboratory data has practically always been there to the point that twenty years ago it was possible to create advanced and extremely sophisticated solutions. Sergio developed furthermore presenting the last 20 years trends in the information exchange technology and how it has evolved from PC based instruments (1985), Client Server Applications (1990) to Web Applications (2005) and new commercial approaches like the SaaS.
He also anticipated that even if so far the driving forces of the Lab informatics journey were the regulations such as FDA, ISO and REACH, those will have to run and propose quickly new regulations, frameworks and adapt to the technology innovation speed. Lately, Sergio has kindly shared Boehringer Ingelheim vision on their future projects and how much they bet on innovative digital solutions.
Even though technology has evolved greatly, the most frequent questions still concern topics about the basis of the laboratory informatics. While Master Data plan is still to be agreed among the DT project team, data silos are building on irrespectively what strategies and systems are implemented. The FAIR principles finally try to give a structure to the catalogs of the data and allow searches that at the beginning of the digitalization of scientific data were absolutely unthinkable. Eric Little, Data officer at Osthus, introduced us to the FAIR principles (Findable, Accessible, Interoperable, Reusable) that is actually more than just an academic idea. They are being applied to help build Data Catalogs, where data is much easier to find, access and integrate across large organizations. Eric showed concrete case studies and explained how advanced statistical methods can be applied to this framework in order to provide advanced analytics and ultimately AI-like capabilities.
The immensity of data opens the door to very important questions about how they are collected, managed, stored and used. Many presentations during plenary and workshops sessions have touched on this delicate point that remains one of the most critical aspects to be addressed in the future.
Pat Pijanowski, Managing Director at Accenture, who, as always, accompanied the participants along a very exciting journey of the lab of the future. He demonstrated with very concrete case studies that if the top five enablers where to be approached individually, they would represent opportunities for lab-centric businesses to innovate rapidly. Approached collectively they would open new pathways into the future. However, despite decades of investment, few companies have achieved fully digital labs and a lot remains to overcome the multiple challenges.
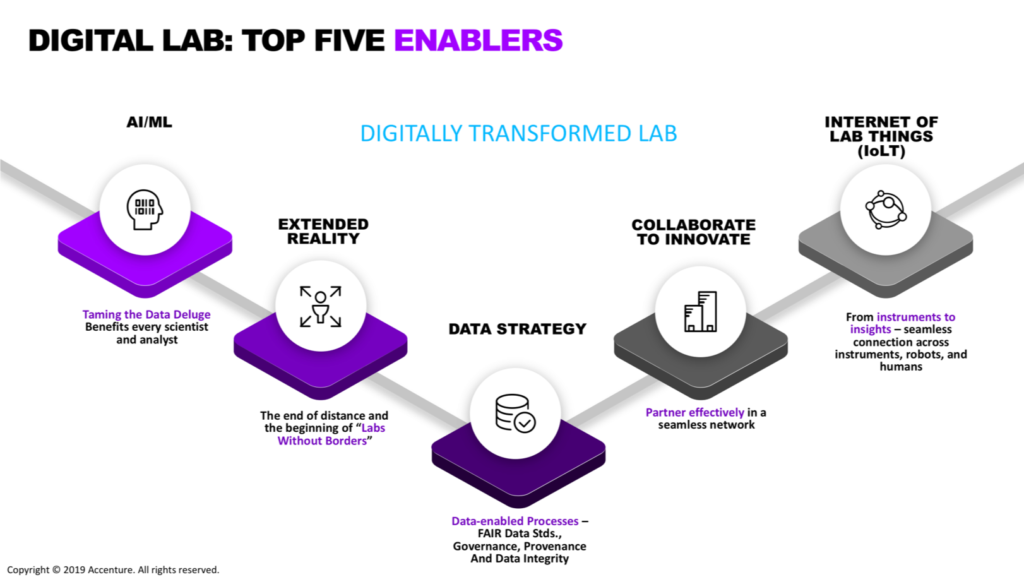
As an immediate consequence, Rik Pepermans, IT Innovation Lead at Unilever, led the participants to ask themselves about the challenges faced during the digital “revolution”. What has emerged is that very often, the human factor and the ability of people to adapt to new technologies remain the most complex aspects to solve.
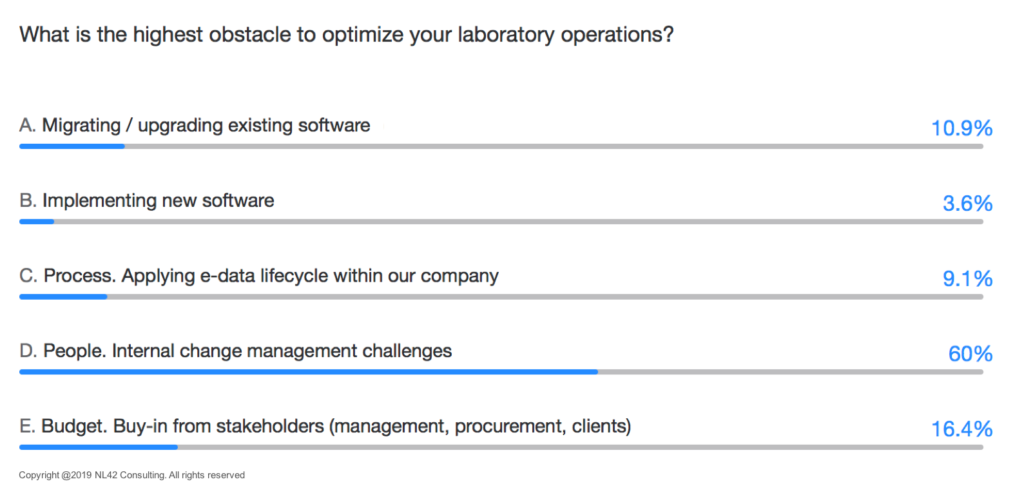
Are we just optimizing or really transforming?
With great pleasure, Gartner has come back attending the seventh edition of the Paperless Lab Academy®. Michael Shanler, Research Vice President at Gartner presented a very concrete path to the digital laboratory and as always, challenged some paradigms about whether the digital transformation is just a digital optimization of the processes rather than a revolutionary and innovative new way of doing.
For sure nowadays enormous amount of data are polled together, analyzed and providing meaningful insights to who is ready to digest and accept them and do something with it. Yet this is about optimizing the business decision making with more solid and verified data points. The transition to digital data management is now essential. Whoever is not ready to it today could be in great difficulty tomorrow. What if the transformation is still about to happen?
Meanwhile, Markus Ruffener, Head of Informatics of Lonza has shared his experience in managing a global enterprise QC Solution upgrade. During his presentation, Markus discussed how to manage a project of this size and focused specifically on the migration and consolidation of large historical data sets coming from multiple instances. From an initial panorama of varied systems and versions, the chosen strategy was to end with a single solution with the objectives to manage the data in a homogeneous, rational and efficient way.
When at NL42 consulting, we´re asked about the real benefits of these projects, we see that the ability to use a consolidated digital platform leads to savings in terms of costs, time, efficiency, qualities that are sometimes underestimated. Clients often ask themselves about tangible benefits to be reached and measured at the end of a journey that appears long and complex. One important aspect that has been emphasized in recent years is the integrity of the data. Very important for the pharmaceutical industry, Data Integrity has become a key issue in every sector, for any data management project involving quality processes. The guarantee that the data produced by the IT systems have been generated, managed and stored appropriately is of fundamental importance for any organization that makes quality a cornerstone of its activities. Fact is that integrity of scientific data is just of sudden attention to all due to the strong focus that the FDA inspectors are putting now, on how data are managed along the product life cycle. How Data Integrity is approached is, in the end, a fair picture of how Quality is approached at the corporate level.
We were therefore very satisfied that an important ISPE representative like Tom De Rudder, Co-chair of the GAMP working group in Europe, attended the congress. Tom has provided an excellent description of the innovations recently introduced in the GAMP guidelines.
As we often say to our customers, the preparation of a project is not always the guarantee of success if the supplier selection process is not followed in strict manners. On many occasions, the commercial presentations of potential suppliers may lead to emotional conclusions and have us missing opportunities to discover a real potential solution for our needs. Peter Cohan, CEO of the Second Derivative company, has spent the last twenty years running GREATDEMO! training courses for suppliers with the aim of improving the way a technical presentation is made. At the Paperless Lab Academy®, Peter accepted the challenge to suggest end-users how to prepare themselves for such technical presentations and ran a presentation that certainly entertained everyone.
Technology and entertainment at the Paperless Lab Academy® 2019
PLA2019 would have not been possible to manage this time without our congress organization going through a digital transformation of our own organizational processes. Not being official event organizers, we had to go through a DT project ourselves and saw the immediate benefits in efficiency, productivity and above all in greater analytics for relevant business decisions making. We decided to repeat the experience in providing a dedicated App to the audience. Participants were able to interact with each other privately and set meetings, access to a digital booklet, take notes, share photos and comments. etc… On many occasions along the two days, presenters have also used the App to ask live questions sharing the participants’ responses in real time. The App once again has been of well accepted and exploited by a larger number of people, meaning that the benefits of networking, interacting and accessing anyway all information type has overcome the desire to hold a full 80 pages booklet. PLA has gone Paperless!
We now look forward to seeing you in 2020. The Paperless Lab Academy® is waiting for you with a double edition, one in Europe – in spring – and one in India – in autumn!
Stay tuned and Follow us on the channel of your choice. The Academy keeps sharing industry news, about upcoming PLA activities, which include webinars and workshops in different locations in Europe.
See you by 2020!
Latest Posts
Key Topics of the PLA2024India
PLA2024India, 5th edition, promises a programme full of interactions and discussions. 4 focused sessions and 2 training workshops The main theme of #P
14 May 2024
Press Release: PLA® Conferences to partner with IA-Meetings for its 5th Indian Edition.
The Paperless Lab Academy® (PLA) is a leading conference about digital transformation of laboratory and quality processes. Above all, it is about mas
08 April 2024
The #PLA2024Europe programme aimed to highlight the importance of the human factor in digital transformation with several presentations and panel disc
19 March 2024
