Operational Excellence QC Lab Benchmark: NL42 to collaborate with University Sant Gallen
NL42 Consulting S.L. has recently signed a collaboration agreement with the University of St. Gallen, Switzerland, on a new Operational Excellence QC Lab benchmarking initiative.
What is the project about?
The Benchmarking exercise helps to assess your Operational Excellence (OPEX) Performance and Maturity in the Quality Control (QC) Lab. The joint development of the benchmarking with leading pharmaceutical companies ensured the right scope of OPEX assessment in QC Labs.
The industry–tested holistic approach of the St. Gallen OPEX Benchmarking ensures a meaningful assessment of your QC Lab taking into account lab specific characteristics.
What are your advantages?
A personalized report shows your relative performance in KPIs in the following categories:
- Productivity
- Quality
- Service
- Cost Efficiency
It further reveals the implementation level of defined Enablers specific to your QC Lab. An extract of a personalized report is available at: http://tectem.ch/institute/opex/qclab-benchmarking
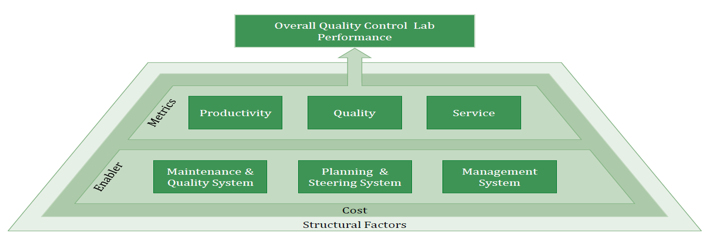
Key focus areas of the benchmarking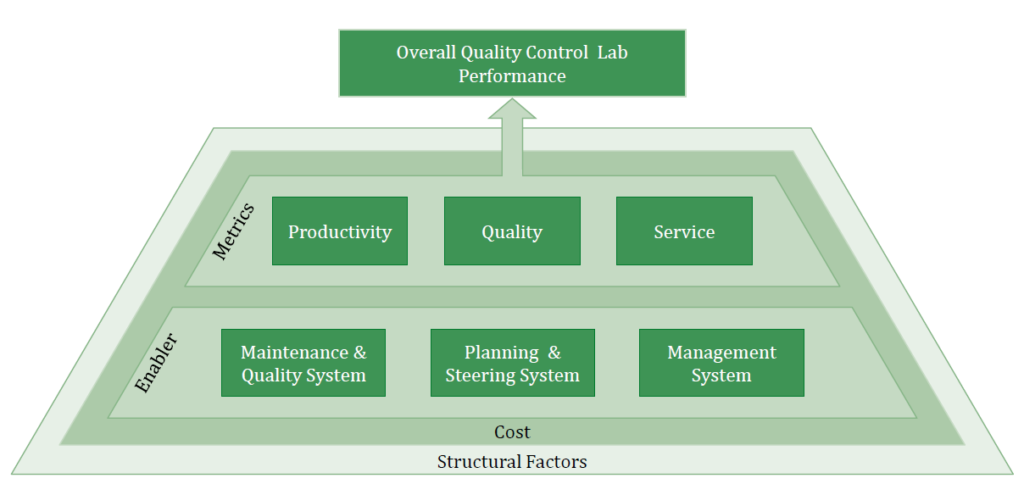
It is important that Operational Excellence is not only measured by performance outcomes but also by analyzing how an organization achieves this performance. To bring performance outcomes and enabling factors together provides a comprehensive picture of your lab’s OPEX Performance and Maturity.
- Productivity: How efficient is the lab doing their job? Disclosed insights on utilization and usage of synergy effects
- Quality: How effective is the lab doing their job? Conclusions about process & product qualit
- Service: Monitor both: Lab processing speed & adherence to schedule
- Maintenance & Quality System: Maintenance, technology assessment, house- keeping, and standardization & simplification.
- Planning & Steering System: Managing commitment & culture, employee involvement & improvement and functional integration
- Management System: Set-up reduction, pull approach, layout optimization, planning adherence and visual management
About the University St. Gallen
USG has large experience in Operational Excellence in the Pharmaceutical Industry and has recently designed a novel QC Lab Benchmarking initiative. www.item.unisg.ch
Professor Thomas Friedli from USG, 10+ years of experience in OPEX in the pharmaceutical industry comments:
“We have seen great interest in this project over the last months by the industry and would like to give the opportunity to any QC Laboratory to participate as well.”
The QC Lab Benchmarking was developed together with the industry to ensure feasibility and usefulness of the outcome. Both KPIs and Enablers make this benchmarking a comprehensive tool to assess the performance and maturity of your QC lab, finally the specific questions regarding structural factors of the lab allows us to compare similar labs to each other.
About NL42
NL42 has large experience in data management at QC laboratories and propose services oriented to paperless processes. www.NL42.com
NL42 sat close to the final users, QC lab mangers and IT partners to review processes and identify data integrity breaches along the whole data life cycle in a laboratory. Afterwhat, NL42 proposes most adequate IT strategy to reach the lab business objectives and and a project plan for implementing lab informatics solutions.
NL42 and the Paperless Lab Academy
NL42 is an independent consultancy company that also organised the European annual congress Paperless Lab Academy. The Congress brings a good opportunity for the industries to interact with opinion leaders, final users and solutions providers.
During the PLA2017, Professor Thomas Friedli has introduced the project and discussed on Key Efficiency Drivers of Quality Control Laboratories in the Pharmaceutical Industry. More information on the St Gallen presentation at PLA2017
Interested in the OPEX QC Lab Benchmark exercise?
Do not hesitate to contact Roberto Castelnovo ( Italian, English, Spanish) or Isabel Munoz-Willery ( French, Spanish, English), both owners at NL42 Consulting.
Roberto.castelnovo@NL42.com | cell: +39 340 456 3204 | skype: nl42_roberto | linkedin
Isabel.munoz@NL42.com | cell: +34 629 026 621 | skype:nl42_isabel | linkedin
Latest Posts
Key Topics of the PLA2024India
PLA2024India, 5th edition, promises a programme full of interactions and discussions. 4 focused sessions and 2 training workshops The main theme of #P
14 May 2024
Press Release: PLA® Conferences to partner with IA-Meetings for its 5th Indian Edition.
The Paperless Lab Academy® (PLA) is a leading conference about digital transformation of laboratory and quality processes. Above all, it is about mas
08 April 2024
The #PLA2024Europe programme aimed to highlight the importance of the human factor in digital transformation with several presentations and panel disc
19 March 2024
