AGILAB WORKSHOP #PLA2019
Unify your GxP and non-GxP laboratories processes to support the complete development cycle
Overview
The nature of laboratory processes and data is highly variable. In some laboratories, the process execution requires flexibility, while in others, processes are executed according to predefined workflows consisting of specific sequences of steps. Current R&D and manufacturing processes are still highly dependent on paper, documents, and spreadsheets.
Under increasing pressure to keep pace with product development and meet the requirements of repeatability, traceability, and efficiency, laboratories are looking for the best solution to digitize their GxP and non-GxP processes.
Agilab-Intelligent Platform
Session Learnings
During this session, attendees will discover how AgiLab Platform will help them to support the complete development cycle (from product batch design and execution, sample management to sample analysis and report of analyses) by:
- Adopting paperless process development activities
- Unifying GxP and non-GxP process executions
- Managing both
- structured data (such as test results, samples, equipment, reagent batches, bill of materials…)
- unstructured data (such as free text, attachments, images and spreadsheet calculations)
- Enabling granular traceability of materials and equipment inventories
- Overcoming the process inefficiencies generated by data silos
- Improving repeatability, collaboration, and data exchange
agilab-process-definition
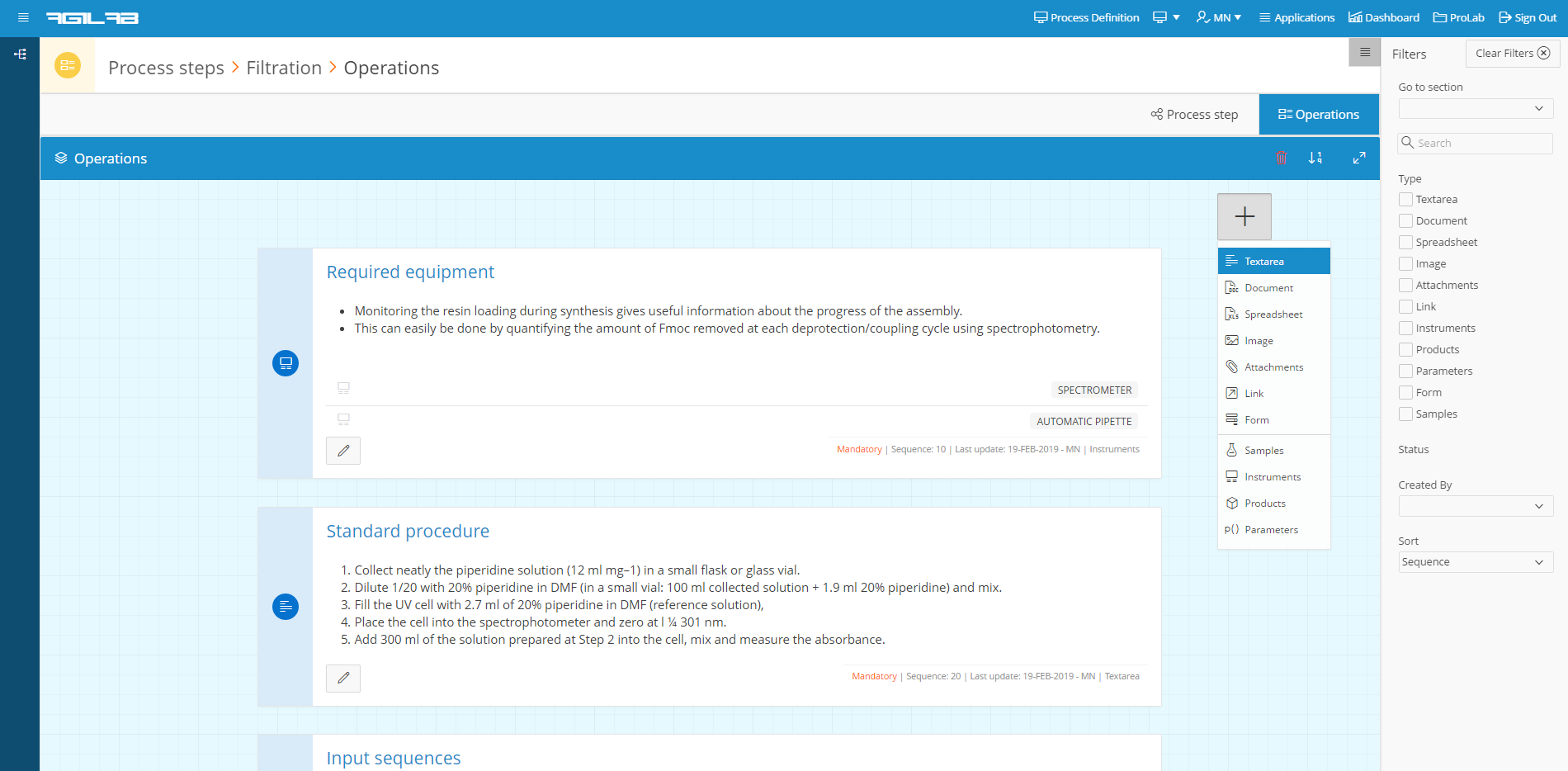
Presenter
AgiLab offers the most advanced unified laboratory digital platform to support operational process from R&D, QA/QC through manufacturing. Built using the latest future-proof web technology powered by Oracle®, AgiLab leverages technologies to help laboratories to accelerate their R&D for faster time to market, improve collaboration across their departments, enhance their decision-making capabilities and meet the increasing regulatory requirements with ease.
During the workshop, AgiLab will present the best-in-class capabilities to move from paper-based to paperless GxP and non-GxP development process workflow.


